
Endometriosis and Adenomyosis: How They Drive Pelvic Pain
A side-by-side guide to how these conditions overlap, differ, and drive pelvic pain.
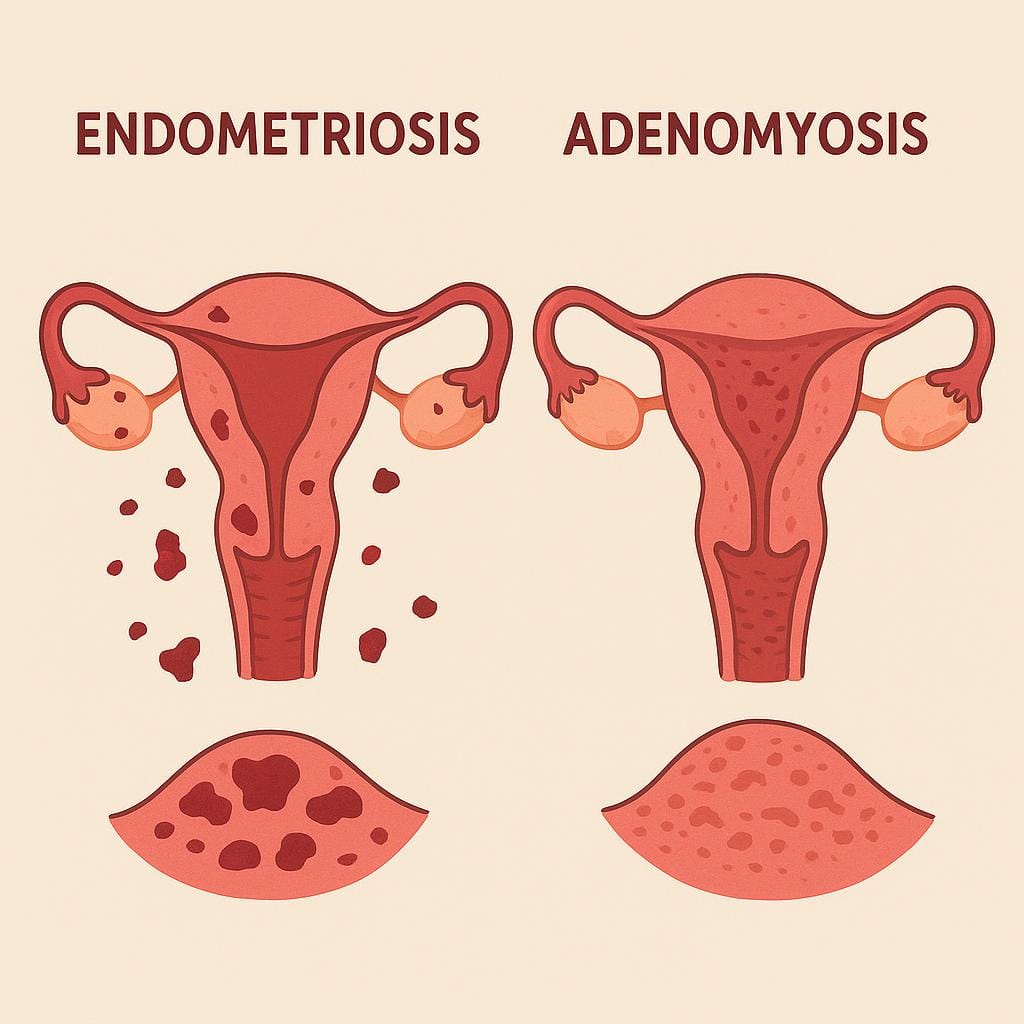
Endometriosis and Adenomyosis: Similarities, Differences, Associations, and Research
Endometriosis and adenomyosis affect millions of women worldwide. While they share certain similarities, they also differ in their pathophysiology, clinical presentation, and management. Comparing and contrasting these conditions helps clarify their overlap, distinctions, and related health associations, with relevant references noted throughout.
Similarities
Both endometriosis and adenomyosis involve the growth of endometrial-like tissue outside the uterine cavity. This ectopic tissue remains responsive to hormonal changes, leading to inflammation, pain, and other similar symptoms that can significantly interfere with quality of life (1). Both conditions predominantly affect women of reproductive age and are associated with dysmenorrhea (painful periods), dyspareunia (painful intercourse), and infertility (2). Although the exact causes are unclear, a combination of genetic, hormonal, and immune factors is thought to contribute to both (3). Either condition can also continue beyond menopause or even be present initially after menopause.
Key Differences
Anatomical location
Although both conditions involve ectopic endometrial-like tissue, they differ in anatomical location. Endometriosis is characterized by endometrial-like tissue outside the uterus, commonly on the ovaries, fallopian tubes, the peritoneum (the pelvic and abdominal skin-like lining), and other organs (4). Adenomyosis, by contrast, is defined by the invasion of endometrial-like tissue into the myometrium, the muscular wall of the uterus (5).
Prevalence
Endometriosis affects approximately 10% of women of reproductive age, whereas adenomyosis is thought to impact between 20% and 35% in this group (6). The true prevalence of both conditions may be underestimated due to the invasive nature of diagnostic procedures and non-specific symptoms (7).
Diagnosis
The gold standard for diagnosing endometriosis is surgery using laparoscopy or robotics, both minimally invasive procedures that allow direct visualization and, if necessary, excision of endometrial-like lesions (8). Adenomyosis, on the other hand, is typically suspected using imaging techniques such as transvaginal ultrasound or magnetic resonance imaging (MRI) and is usually confirmed by a pathologist when the uterus is removed (9). An accurate preoperative biopsy is very difficult; however, when adenomyosis is not diffuse throughout the myometrium, discrete adenomyomas can sometimes be removed while leaving the uterus in place.
Treatment
Both conditions are managed with a combination of medical and surgical therapies tailored to symptom severity and reproductive goals. Hormonal therapies—including oral contraceptives, progestins, and gonadotropin-releasing hormone (GnRH) agonists and antagonists—are commonly used to manage symptoms in both endometriosis and adenomyosis (10). Integrative measures, including anti-inflammatory and anti-oxidant hormone-modulating nutrition and lifestyle modifications, can help control symptoms and may contribute to treating root causes. Surgical approaches differ by condition. In endometriosis, the preferred intervention is laparoscopic or robotic excision of ectopic tissue (11). For adenomyosis, hysterectomy (removal of the uterus) may be considered in severe cases when fertility preservation is not a concern (12). In selected cases where imaging identifies discrete adenomyomas, removal while preserving the uterus is possible; the decision to remove the uterus is highly individualized.
Struggling with Pelvic Pain? Get Help Now
Our specialists are here to help you understand your condition and explore your treatment options.
Schedule Your VisitCoexistence and Associated Conditions
Endometriosis and adenomyosis can coexist in the same patient, and one study found that adenomyosis is significantly more prevalent among women with endometriosis (13). Coexistence may exacerbate symptoms and complicate both diagnosis and management (14). These conditions are also linked to other health issues. Women with either may experience chronic pelvic pain that can be debilitating and significantly impact daily life (15). Adenomyosis and uterine fibroids (leiomyomas) may occur together, which can further complicate diagnosis and treatment despite being distinct entities (16). Women with endometriosis have an increased risk of autoimmune and inflammatory disorders—such as rheumatoid arthritis, systemic lupus erythematosus, and inflammatory bowel disease—while similar associations with adenomyosis are less well-established but have been suggested in some studies (17, 18). Mental health concerns, including depression, anxiety, and decreased quality of life due to chronic pain and infertility, are linked to both conditions (19).
Research and Future Directions
There is a growing body of research targeting the pathophysiology, diagnosis, and treatment of endometriosis and adenomyosis. Key areas include the identification of specific biomarkers to improve diagnostic accuracy and enable earlier intervention (20), advances in non-invasive imaging techniques to reduce reliance on invasive diagnostic procedures (21), exploration of novel therapeutic approaches such as targeted hormonal therapies, immunomodulators, and anti-inflammatory agents to improve symptom control and fertility outcomes (22), and investigation of genetic and epigenetic factors that drive development and progression to inform future treatment strategies (23).
References
Vercellini P, Viganò P, Somigliana E, Fedele L. (2014). Endometriosis: pathogenesis and treatment. Nat Rev Endocrinol. 10(5): 261-75. DOI: 10.1007/s10735-025-10499-z
Parazzini F, Esposito G, Tozzi L, Noli S, Bianchi S. (2017). Epidemiology of endometriosis and its comorbidities. Eur J Obstet Gynecol Reprod Biol. 209: 3-7. DOI: 10.1016/j.rbmo.2025.105102
Zondervan KT, Becker CM, Koga K, Missmer SA, Taylor RN, Viganò P. (2018). Endometriosis. Nat Rev Dis Primers. 4(1): 9.
Giudice LC, Kao LC. (2004). Endometriosis. Lancet. 364(9447): 1789-99. DOI: 10.1007/s00210-025-04935-w
Vannuccini S, Tosti C, Carmona F, Huang SJ, Chapron C, Guo SW, Petraglia F. (2017). Pathogenesis of adenomyosis: an update on molecular mechanisms. Reprod Biomed Online. 35(5): 592-601. DOI: 10.1016/j.rbmo.2017.06.016
Garcia L, Isaacson K. (2011). Adenomyosis: review of the literature. J Minim Invasive Gynecol. 18(4): 428-37. DOI: 10.3390/jcm14051430
Chapron C, Marcellin L, Borghese B, Santulli P. (2019). Rethinking mechanisms, diagnosis and management of endometriosis. Nat Rev Endocrinol. 15(11): 666-82. DOI: 10.1038/s41574-019-0245-z
Johnson NP, Hummelshoj L, Adamson GD, Keckstein J, Taylor HS, Abrao MS, et al. (2017). World Endometriosis Society consensus on the classification of endometriosis. Hum Reprod. 32(2): 315-24. DOI: 10.1093/humrep/dew293
Champaneria R, Abedin P, Daniels J, Balogun M, Khan KS. (2010). Ultrasound scan and magnetic resonance imaging for the diagnosis of adenomyosis: systematic review comparing test accuracy. Acta Obstet Gynecol Scand. 89(11): 1374-84. DOI: 10.3109/00016349.2010.512061
Vercellini P, Buggio L, Berlanda N, Barbara G, Somigliana E, Bosari S. (2016). Estrogen-progestins and progestins for the management of endometriosis. Fertil Steril. 106(7): 1552-71.e2. DOI: 10.1080/14728214.2023.2296080
Yeung P Jr, Sinervo K, Winer W, Albee RB Jr. (2011). Complete laparoscopic excision of endometriosis in teenagers: is postoperative hormonal suppression necessary? Fertil Steril. 95(6): 1909-12, 1912.e1. DOI: 10.1016/j.fertnstert.2011.02.037
García-Solares J, Donnez J, Donnez O, Dolmans MM. (2018). Pathogenesis of uterine adenomyosis: invagination or metaplasia? Fertil Steril. 109(3): 371-9. DOI: 10.1158/2159-8290.CD-25-0778
Mijatovic V, Florijn E, Halim N, Schats R, Hompes P. (2010). Adenomyosis has no adverse effects on IVF/ICSI outcomes in women with endometriosis treated with long-term pituitary down-regulation before IVF/ICSI. Eur J Obstet Gynecol Reprod Biol. 151(1): 62-7. DOI: 10.1016/j.ejogrb.2010.02.047
Pinzauti S, Lazzeri L, Tosti C, Centini G, Orlandini C, Luisi S, et al. (2015). Coexistence of endometriosis and adenomyosis in women with chronic pelvic pain. J Obstet Gynaecol Res. 41(6): 909-14. PMID: 23610949
Howard FM. (2003). Chronic pelvic pain. Obstet Gynecol. 101(3): 594-611. DOI: 10.1016/j.jmig.2026.01.014
Stewart EA. (2015). Uterine fibroids. Lancet. 387(10022): 1189-99. DOI: 10.1080/13625187.2026.2615974
Sinaii N, Cleary SD, Ballweg ML, Nieman LK, Stratton P. (2002). High rates of autoimmune and endocrine disorders, fibromyalgia, chronic fatigue syndrome and atopic diseases among women with endometriosis: a survey analysis. Hum Reprod. 17(10): 2715-24. DOI: 10.1093/humrep/17.10.2715
Benagiano G, Brosens I, Habiba M. (2015). Structural and molecular features of the endomyometrium in endometriosis and adenomyosis. Hum Reprod Update. 21(4): 445-58. DOI: 10.1093/humupd/dmt052
Roomaney R, Kagee A. (2016). The association between pain, disability, fatigue and depression in women diagnosed with endometriosis: a moderated mediation analysis. J Psychosom Obstet Gynaecol. 37(4): 137-44.
Nisenblat V, Bossuyt PM, Shaikh R, Farquhar C, Jordan V, Scheffers CS, et al. (2016). Blood biomarkers for the non-invasive diagnosis of endometriosis. Cochrane Database Syst Rev. 5: CD012179. DOI: 10.3390/healthcare13243276
Brosens I, Gordts S, Campo R, Benagiano G. (2016). Non-invasive methods of diagnosis of endometriosis. Curr Opin Obstet Gynecol. 28(4): 267-76. DOI: 10.3390/jcm15010030
Stratton P, Berkley KJ. (2011). Chronic pelvic pain and endometriosis: translational evidence of the relationship and implications. Hum Reprod Update. 17(3): 327-46. DOI: 10.1093/humupd/dmq050
Zondervan KT, Rahmioglu N, Morris AP, Nyholt DR, Montgomery GW, Becker CM, et al. (2016). Beyond endometriosis genome-wide association study: from genomics to phenomics to the patient. Semin Reprod Med. 34(4): 242-54. DOI: 10.1055/s-0036-1585408
Quick Answers
How rare is endosalpingiosis?
Endosalpingiosis is generally considered uncommon, but “how rare” it is depends heavily on who’s being studied and how it’s found. Many cases are discovered incidentally on pathology—meaning tissue is identified under the microscope after surgery done for other reasons—so it’s likely underrecognized in the general population. In other settings (like surgical cohorts), it may appear more often simply because more tissue is being sampled and examined carefully.
What matters most for patients is that endosalpingiosis can be confused with endometriosis on imaging or even at surgery, yet it doesn’t always behave the same way clinically. If you’ve been told you have endosalpingiosis and you also have pelvic pain, bowel/bladder symptoms, or fertility concerns, our team can help interpret what that finding means in the context of your symptoms and operative/pathology reports. You’re welcome to explore our educational content on related endometriosis and uterine conditions, and reach out to schedule a consultation if you want a personalized plan.
Can endometriosis cause a painful bump near the anus?
Yes. Endometriosis can contribute to pain and pressure around the rectum and anal area, especially when disease involves the rectum/rectosigmoid region or nearby tissues. Many patients describe deep pain with bowel movements, rectal pressure, or symptoms that flare around their cycle, and those patterns can fit bowel or deep infiltrating endometriosis.
That said, a sensitive bump on the anus itself is more often something else (like a hemorrhoid, fissure, skin infection/abscess, or another localized anal/skin condition). In some cases, pelvic disease can coexist with these issues, which is why we don’t assume every finding is endometriosis—or dismiss it as “nothing.”
If you’re noticing a new, persistent, or worsening bump—especially if it’s very tender, draining, bleeding, or associated with fever—we want to evaluate the full picture. Our team can sort out whether your symptoms point toward bowel endometriosis, a separate anorectal condition, or both, and plan next steps such as a focused exam and, when appropriate, expertly interpreted imaging to map possible deep disease.
When is menstrual bleeding considered too heavy?
Menstrual flow is generally “too heavy” when it consistently disrupts your life or overwhelms your usual period products—think flooding or soaking through pads/tampons quickly, passing frequent or large clots, needing to double up, or bleeding long enough that you can’t plan around it. Another major clue is fatigue, dizziness, or shortness of breath that can come with iron deficiency from ongoing blood loss. If you’re timing your day around bathrooms, waking at night to change products, or avoiding work, exercise, travel, or sex because of bleeding, that’s not something we consider “normal.”
Heavy bleeding is a symptom, not a diagnosis, and common underlying drivers include adenomyosis, fibroids, hormonal imbalance, and sometimes endometriosis—especially when heavy bleeding shows up with severe cramps or deep pelvic pain. Because imaging and symptoms don’t always match (a scan can look “mild” while symptoms are intense), we take a symptom-led approach and look at the full pattern, including pain, pressure, clots, cycle timing, and any signs of anemia. If your bleeding feels like it’s escalating or you’ve been told to “just live with it,” our team can help you sort out likely causes and build a plan that targets the source—not just the bleeding.
Can endometriosis cause arthritis-like joint pain?
Yes—endometriosis can be associated with arthritis-like joint pain in some people, even though joint pain isn’t considered a classic “core” symptom. Endometriosis can drive chronic inflammation and immune dysregulation, and that whole-body inflammatory state may show up as aching, stiffness, or flares that feel similar to inflammatory arthritis. Some patients also notice joint symptoms that cycle with their period or worsen during broader endometriosis flares.
At the same time, endometriosis doesn’t “equal” autoimmune arthritis, and an association doesn’t prove that one causes the other. Research suggests higher rates of certain autoimmune conditions in people with endometriosis—including inflammatory diseases that can affect joints—so persistent joint pain deserves a full-picture evaluation rather than being automatically attributed to pelvic disease alone. If you’re dealing with pelvic pain plus joint symptoms, our team can help you sort out what fits endometriosis, what may be a related immune condition, and how that affects your treatment plan, including whether excision surgery and coordinated integrative support make sense for you.
How does estrogen affect the endometrium?
Estrogen is one of the main hormones that drives endometrial growth. In the first half of the menstrual cycle, rising estrogen signals the endometrium to thicken and rebuild after a period, preparing the uterus for a possible pregnancy. It also influences the local immune and inflammatory environment in the uterus, which is part of why hormonal shifts can change bleeding patterns and pain.
When estrogen’s growth signals are strong—and progesterone’s “calming” effect is weaker than expected (often described as progesterone resistance)—the endometrium can behave in a more persistently inflamed, reactive way. This hormone–inflammation pattern is especially relevant in estrogen-dependent conditions like adenomyosis and endometriosis, where tissue similar to the endometrium can contribute to ongoing symptoms. If you’re trying to make sense of heavy bleeding, severe cramping, or cycle-linked pelvic pain, our team can help you connect the hormonal biology to what you’re feeling and review next steps for diagnosis and treatment.

